How do we extract physiologically meaningful quantities from noisy optical signals – reliably enough to trust across time, individuals, and clinical settings?
Research Themes
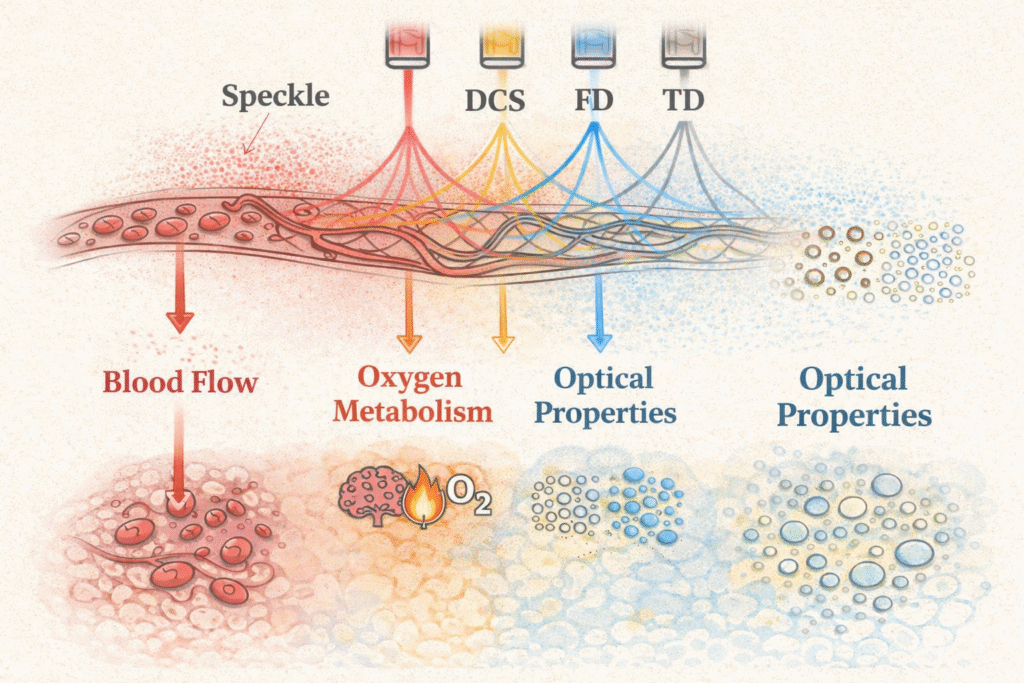
Physiologically interpretable diffuse optics
Most optical neuroimaging produces oxygenation signals: a convolution of blood flow, oxygen metabolism, and blood volume that is difficult to interpret. I develop methods to access more interpretable quantities: cerebral blood flow via DCS, oxygen metabolism via combined DOS/DCS, and absolute optical properties via TD- and FD-DOS. Work here includes forward and inverse light-transport modelling, calibration frameworks, and the theoretical limits of what near-infrared light can encode in heterogeneous tissue.
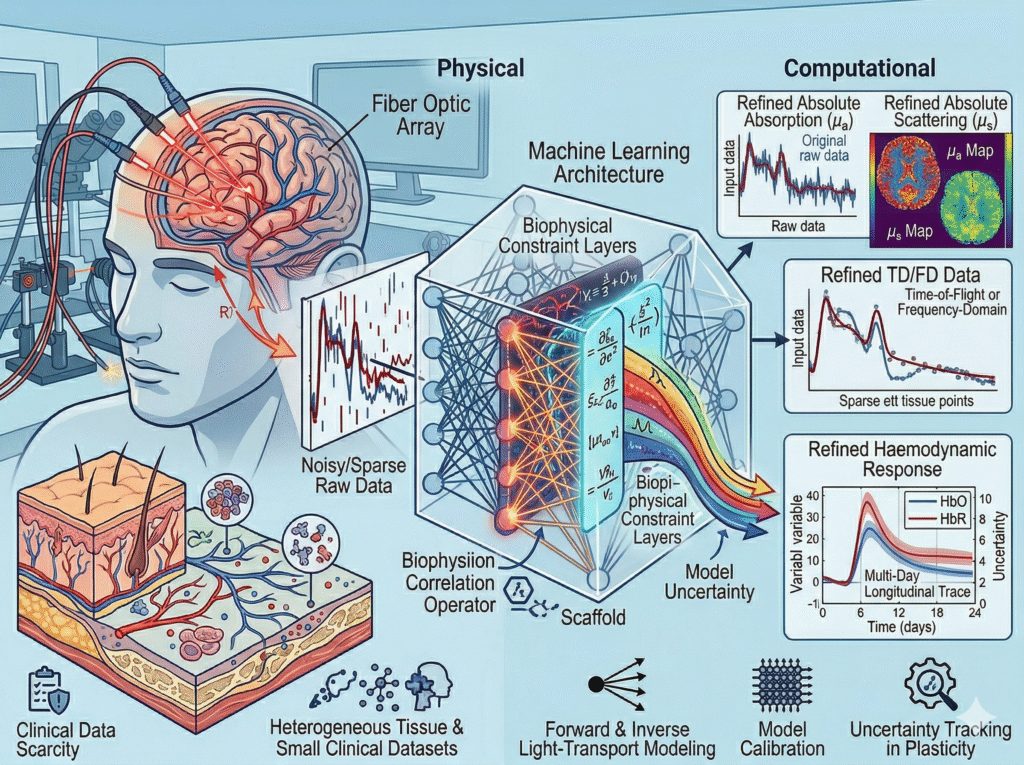
Physics-informed machine learning for optical neuroimaging
Data-driven methods alone are poorly suited to optical neuroimaging: clinical datasets are small, tissue is heterogeneous, and physically implausible solutions are a genuine mistake. Work here develops machine learning architectures constrained by models. Applications include signal denoising, optical property estimation from noisy or sparse data, and haemodynamic response modelling in fNIRS. A central question is how to propagate model uncertainty – not just data uncertainty – through the inference pipeline.
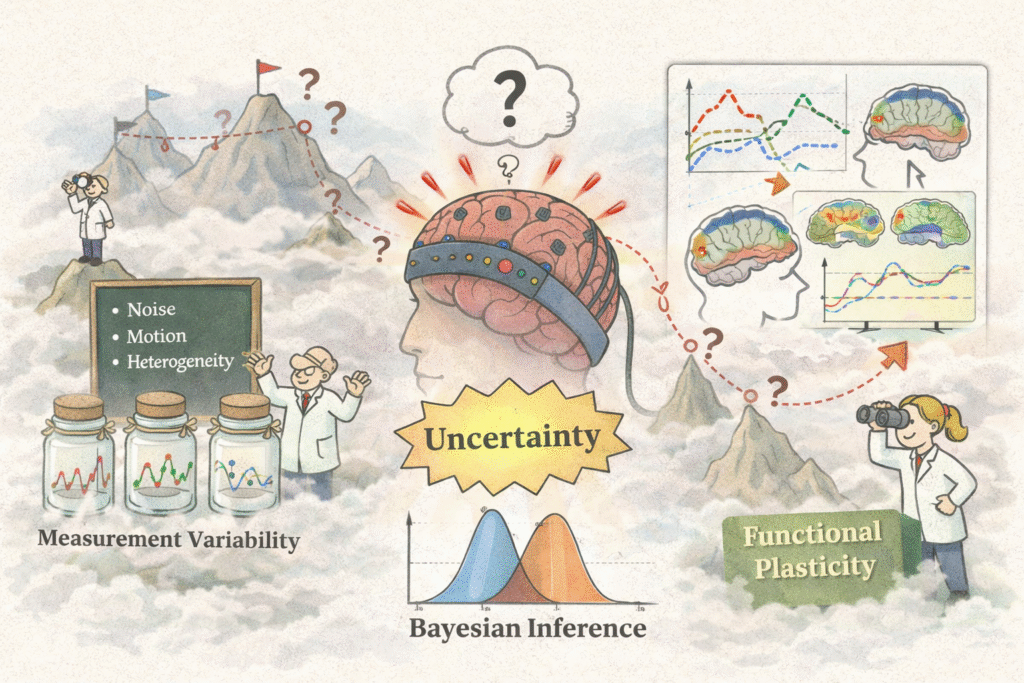
Uncertainty quantification and reproducibility by design
Trustworthy measurement requires knowing not just what a quantity is but also how confident we should be in that estimate. I develop Bayesian inference frameworks for DCS and fNIRS time series, methods for decomposing and reducing sources of variability across sessions and individuals, and protocol strategies that treat reproducibility as a quantitative requirement rather than a validation afterthought. The goal is longitudinal monitoring and plasticity tracking that is statistically meaningful.
Brain health, complexity, and longitudinal dynamics
The methods developed are tested and motivated by real questions about the brain over time. A central thread is the brain’s complexity and network organisation: how functional connectivity and dynamic network topology change with injury, disease, and recovery, and what those changes reveal about underlying physiology. Using fNIRS and DCS, my group studies brain-body interactions and cerebrovascular dynamics at the mesoscopic scale – the level where systems neuroscience meets physiology and where slow biological change becomes detectable only with reliable longitudinal methods.
What is Diffuse Optics?
Light has long been used for characterizing a broad spectrum of samples. One of the simplest techniques relies on light absorption, which provides information on the presence and concentration of molecules in a sample based on their specific absorption characteristics. Light scattering experiments reveal sample morphology, including the size and refractive indices of scatterers, as well as the structural organization of scatterer collections. Techniques such as dynamic or quasi-elastic light scattering provide insight into the motion of scatterers by analyzing temporal variations in scattered light intensity. These optical spectroscopic techniques are well-established for simple, homogeneous, and optically thin samples.

Diffuse optics focuses on light propagation in highly scattering media (also called turbid media), where scattering dominates over absorption. Such media are can be found in various fields, including biology, environmental science, and atmospheric studies. For example, colloids, which are suspensions of particles in a fluid, scatter light significantly and are a common subject of study in diffuse optics. Near-infrared light (~700-900 nm) is also extensively scattered by biological tissues, due to their heterogeneity. Similarly, environments like soil, water, and atmospheric conditions contain particles and impurities that scatter light. Diffuse optics provides a framework for understanding and predicting light behavior in these complex, inhomogeneous media.

Due to high scattering, light propagation in turbid media exhibits a diffusion profile such as the one shown in the figure. Modeling light propagation in diffusive media can be approached through deterministic analytical or computational models, as well as stochastic methods like Monte Carlo simulations. Regardless of the approach, these models have one common goal: given the optical properties of the medium such as absorption and scattering coefficients, or the mean-square displacement of the moving scatterers in tissue, they aim to estimate observable experimental quantities, such as the light intensity or the speckle-based pattern detected at the medium’s surface.
Diffuse optical techniques use these measured optical quantities to recover the optical properties of a diffusive medium. These experimental techniques can be broadly classified into static and dynamic methods. Static methods are commonly referred to as near-infrared spectroscopy (NIRS) or diffuse optical spectroscopy (DOS) and focus on slow variations in absorption and scattering. Dynamic methods encompass diffuse correlation spectroscopy (DCS), laser speckle imaging (LSI), speckle-contrast optical spectroscopy (SCOS), among others, and they aim to monitor speckle fluctuations in scattered light, which are sensitive to the motion of scatterers.
Both static and dynamic diffuse optical methods have been widely applied in clinical and neuroscientific research. These techniques are noninvasive, portable, cost-effective, and suitable across the human lifespan, making them attractive alternatives to traditional neuroimaging modalities. My research focuses on developing analytical and computational models to improve the quantification of these optical methods and on evaluating their potential as biomarkers of brain health and disease through statistical learning and artificial intelligence.
